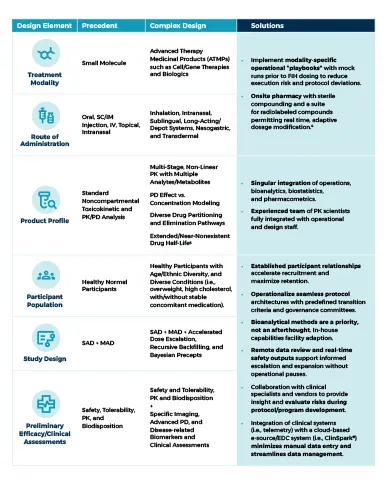
Simple First-in-Human (FIH) study designs are increasingly giving way to complex, multifaceted programs that address sophisticated hypotheses early in clinical development. Today’s sponsors demand traditional safety and pharmacokinetic assessments along with early evidence of target engagement and clinical efficacy. We are seeing the emergence of innovative “hybrid” study designs that integrate both healthy volunteers and patients while navigating advanced modalities, diverse endpoints, and accelerated timelines.
Read our infographic to see how differentiated clinical pharmacology units adapt to meet these evolving FIH challenges.