
The science of cancer care has improved to the extent that survival alone does not fully capture all elements of a successful treatment, prompting a pivotal shift towards integrating patient-reported outcomes (PROs) in oncology clinical trials. These outcomes reach beyond providing simple metrics; they express the voices and experiences of patients, offering an invaluable perspective that resonates across regulatory bodies, payors, and patients themselves.
Below, we delve into how PROs enrich the oncology field by enhancing regulatory decisions, refining payor assessments, and, most importantly, improving patient care. You can read a detailed discussion in our white paper.
Patient Reported Outcomes in Oncology Clinical Trials
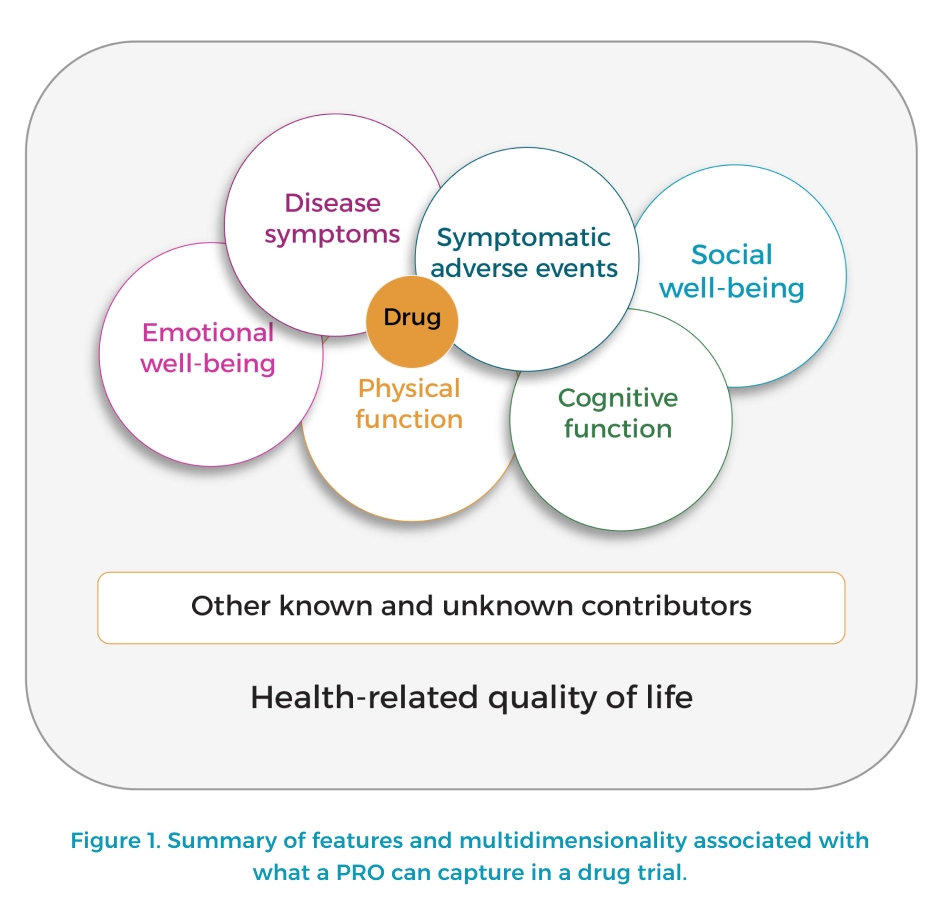
Patient-reported outcomes are measurements derived directly from patients, reflecting individuals’ perspectives on their health and the impacts of treatment and care on quality of life. These outcomes include various domains such as physical symptoms, emotional well-being, social functioning, and overall health-related quality of life (hrQOL). In the context of oncology, PROs are particularly crucial as they provide insights into the effects of cancer and its treatment from the patient’s viewpoint, offering a comprehensive assessment of treatment efficacy, tolerability, and the overall burden of the disease.
By integrating PROs into clinical trials and care strategies, healthcare providers can better understand the full impact of cancer therapies, thus enabling more patient-centered care and informed clinical decisions. Including PRO data on drug labels also allows patients to understand their treatment options from the perspective of other patients. Perhaps in recognition or reflection of the growing need for oncology PROs, global regulatory bodies are pushing toward implementing PROs as a necessity for cancer drug approval.
The Growing Importance of PROs for Regulatory Bodies
Regulatory agencies like the FDA and the EMA have increasingly recognized the significance of PROs. These bodies have established guidelines that emphasize the inclusion of PROs in clinical trials, ensuring that any assessment of a new therapy extends beyond traditional clinical outcomes and considers its impact on the patient’s quality of life.
For instance, the FDA’s guidance for the industry on core patient-reported outcomes in cancer clinical trials highlights the need for these measures in evaluating the broader benefits and risks associated with cancer therapies. In another FDA guidance communication, Patient-Reported Outcome Measures: Use in Medical Product Development to Support Labeling Claims, there is an important dialogue around establishing clinically meaningful improvements in patient-reported symptoms, presenting PROs as an efficacy endpoint. Beyond regulatory initiatives, incorporating PROs can increase the “value” of your therapeutic from a payor perspective, ultimately helping formulary placement and reimbursement post-approval.
PROs Offer Strategic Value to Payors

Getting a cancer therapeutic approved is one hurdle, but regulatory approval does not directly govern patient access. Instead, payors control access (e.g., governmental and commercial insurance providers) based on which drugs they decide to cover or reimburse. For payors, PROs offer a robust tool for performing cost-effectiveness analyses and making informed reimbursement decisions. By quantifying how treatments affect patients’ daily lives and long-term health, payors can gauge the true value of medications beyond traditional clinical efficacy outcomes. PROs help paint a more detailed picture of a drug’s overall impact, aiding payors in creating comprehensive care models that are both informative and economically sustainable.
Based on published payor surveys, the following PRO factors have the greatest impact on the decision-making process in evaluating a cancer therapeutic for reimbursement:
- High-quality, well-managed design
- Clear PRO evidence
- Psychometrically validated PRO measures
- PRO publication in reputable peer-reviewed journals
PROs Are the Future of Cancer Care
PROs are reshaping oncology research and treatment by aligning the goals of regulatory bodies, payors, and healthcare providers with patients’ needs and experiences. As we advance, the continued adoption and refinement of PRO methodologies will be crucial in developing therapies that not only combat disease but also enhance the quality of life for those receiving them.
Incorporating PROs into clinical practice also presents obstacles, warranting a detailed review of the complexity of data collection, patient privacy concerns, and the need for standardized measurement instruments. In addition, there are further nuances for multi-regional clinical trial designs, as well as phase-dependent factors. Read our full white paper for more insights.
There is no one-size-fits-all approach for employing PROs. Explore the transformative impact that PROs are having on clinical research, consider how to position these outcomes with various stakeholders, and understand how to integrate them into your research frameworks to better serve patients and streamline the drug development processes — contact us today to set up a time to discuss how we can be your PRO partner to custom-tailor the best PRO strategy for your next oncology study!