
Adaptive study designs (ASD) increasingly are vetted for use across a wide range of therapeutic indications, particularly for orphan disease indications. The resurgence of interest in innovative clinical trial methodology is born of need for efficient and informed program development and study design suitable for investigation of unique products, in unique therapeutic indications, in which benefits associated with traditional clinical trial methodology are not applicable.

Many orphan diseases have disease pathways that result in diverse expressions of pathology, requiring multiple clinical assessments for comprehensive evaluation of interventional products targeting orphan disease pathology. For example, dose response relationships are often uncertain; differences in genetic and phenotypic expression must be accounted for in small patient samples; and unlike many traditional therapeutic indications, there is little opportunity to replicate clinical study results given the small populations of patients that are affected by orphan diseases. Additionally, these diseases often affect children, many times with devastating consequences. Thus, efficient and informed product evaluation, which nevertheless adheres to good standards of methodological rigor, becomes a mandatory asset.
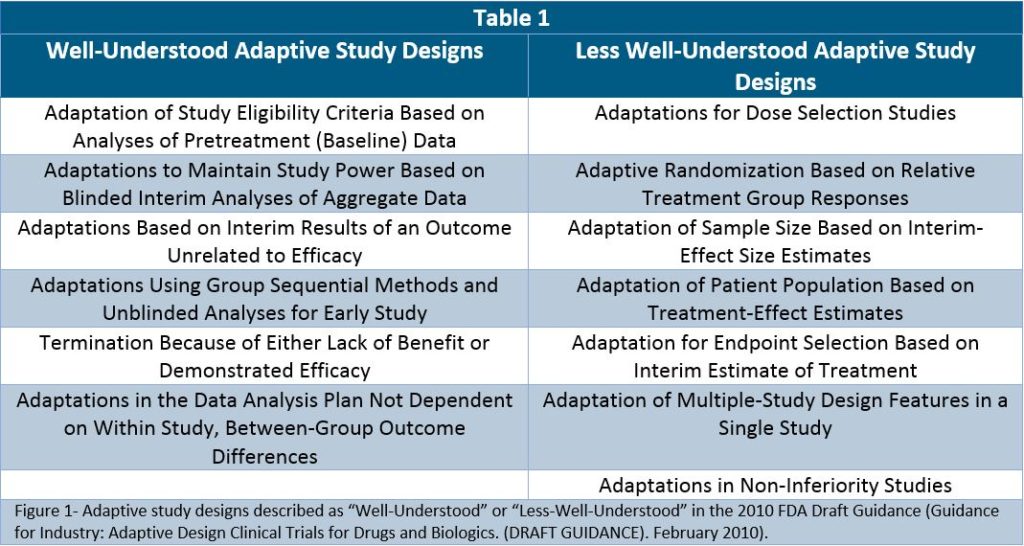
Herein lies the promise of adaptive study designs for orphan diseases, which allow for greater flexibility in product evaluation and shorter overall program development timelines, while still achieving the highest scientific integrity under a sanctioned regulatory umbrella. Representative ASDs are depicted in Table 1 below. Furthermore, as interest has increased in ASDs so too has knowledge of how to execute these clinical programs, which by definition are frequently associated with operational complexity because of the many adaptations which ensue.
Combining adaptive design models represents the next frontier in clinical trial design innovation
For example, the 2010 FDA guidance on adaptive study designs classified designs into “generally well-understood adaptive designs with valid approaches to implementation” or “adaptive study designs whose properties are less well understood” (Table 1). This demarcation reflected uncertainty within the biostatistical and regulatory community regarding methods of analysis and interpretation, but in many respects it also mimicked the operational challenges attendant to many of these trial designs. However, though not yet reflected in updated regulatory guidance, these design permutations were discussed at the 2018 Adaptive Clinical Trial Symposium in Philadelphia, with attendees noting that most are now considered “well-understood” by the field, including biostatistical reviewers at the FDA, and that the challenge now is how best to combine these models for even greater innovation in ASD designs.
Case Studies: ASD Development in Orphan Disease Program
Within the last 12 months, Worldwide Clinical Trials has been instrumental in five adaptive study designs for orphan diseases, which all have a two-stage component permitting adaptation in dose (dropping uninformative dosages), or eligibility criteria (shaping eligibility criteria to enhance inclusion of more patients with likely response). These designs have occurred in orphan indications within central nervous system disorders, hematological disorders, and rheumatologic conditions. Notably, none of the clinical studies were in oncology, a therapeutic area which has pioneered the use of these designs, – an observation reflecting the breakthrough occurring across therapeutic areas which now routinely exploit these approaches.
“Building the bridge slowly to cross it quickly”
For all programs, both traditional (phase 1, 2, and 3 studies) and adaptive design (seamless phase 2/3 designs) were vetted for feasibility highlighting costs, timelines, impact on patient recruitment and retention, sensitivity of proximal and distal endpoints, and corporate goals. These factors required extensive discussion among various stakeholders (sponsor, trial feasibility specialists, KOLs, operation specialists, statistical experts) to develop an extensive pro-and-con assessment for each program highlighted in the Figure 1 below.
Although there are many permutations, all adaptive study designs which have been utilized by Worldwide have used a two-stage, inferentially or operationally seamless phase 2/3 approach. The two-stage design optimizes the treatment dose selection process, as well as allows for determination of clinical efficacy within one protocol (Stage 1 – dose optimization/safety and Stage 2 – determination of efficacy/safety), thus addressing the requirement for dose selection and efficacy in a small sample of orphan disease patients. Additionally, as uninformative doses are discontinued early during the clinical trial based upon accruing efficacy and safety data, more patients are exposed to a potentially optimal dose compared to alternative approaches.

Academic, sponsor, CRO collaboration
Because of its complexity, the adaptive study design process includes team members who must be familiar with the pharmacological attributes of the investigational agent (sponsors) biostatistical staff (implications of using an inferentially seamless vs. operationally seamless design); scientific and medical input (identification of interim biomarkers/endpoints with KOL interface); operations team (trial execution, organization of stages); clinical assessments technologies (identification of relevant endpoints); and members from bids and proposals (budgets, timelines). Although team composition is constant, leadership within each ASD process will change contingent upon the phase of development and clinical study objectives. Thus, the process of ASD development is ultimately one of adjudication in which the needs of different stakeholders merge into a coherent concept, ideally suited to orphan indications and the innovative technology to treat them.
Learn More about Our Expertise in Orphan Disease Clinical Trials
To learn more about Worldwide Clinical Trials’ expertise in orphan disease clinical research, visit our rare disease/orphan drug trials page. If you’re looking for a CRO partner for your next orphan disease clinical trial, please provide a request for proposal.
Reference
1. FDA. Guidance for Industry: Adaptive Design Clinical Trials for Drugs and Biologics. (DRAFT GUIDANCE). February 2010